For children 1 year and older with BRAF V600E+ low-grade glioma,
Transform what's possible with TAFINLAR + MEKINIST
TAFINLAR + MEKINIST is known as a targeted therapy (or treatment). This means that it is designed to specifically control the abnormal BRAF V600E mutation and slow the growth of cancerous cells. TAFINLAR + MEKINIST is a combination therapy of 2 medications that work together to target and treat the tumor. | |
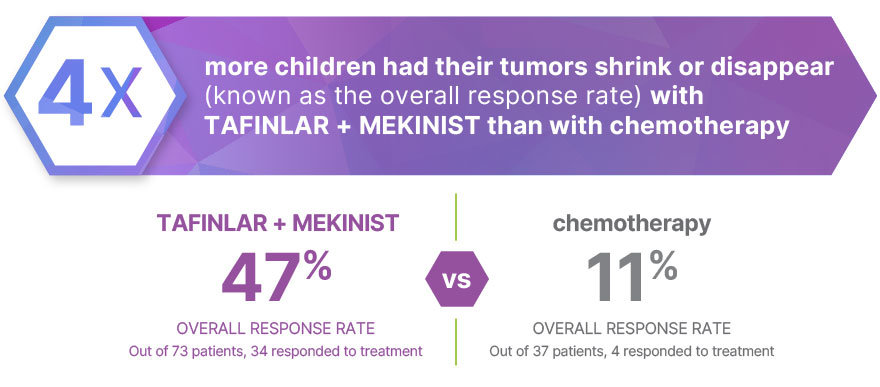
Research shows that more children have a reduction in tumor size with TAFINLAR + MEKINIST than with chemotherapy (vincristine and carboplatin). |
What are the benefits of TAFINLAR + MEKINIST?
In a clinical trial:
Tumors disappeared completely in 3% (2 out of 73) of patients treated with TAFINLAR + MEKINIST and 3% (1 out of 37) of patients treated with chemotherapy
Tumors shrank in 44% (32 out of 73) of patients treated with TAFINLAR + MEKINIST and 8% (3 out of 37) of patients treated with chemotherapy
TAFINLAR + MEKINIST is an FDA-approved treatment that was researched in 110 children with BRAF V600E+ low-grade glioma (LGG) whose cancer returned after surgery or was not able to be removed by surgery.
110 children → 73 treated with TAFINLAR + MEKINIST, 37 treated with chemotherapy (vincristine and carboplatin)
Does TAFINLAR + MEKINIST have side effects?
Serious side effects
For a complete list of potential side effects, please see “Important Safety Information" below. Serious side effects associated with TAFINLAR + MEKINIST include risk of new skin cancers, bleeding problems, inflammation of the intestines or tears (perforation) of the stomach or intestines, blood clots, heart problems, eye problems, lung or breathing problems, fever, serious skin reactions, increased blood sugar, a type of anemia called hemolytic anemia, and hemophagocytic lymphohistiocytosis.
TAFINLAR + MEKINIST has side effects that you should be aware of. The most common side effects seen when using TAFINLAR + MEKINIST in children with LGG are:
Fever
Rash
Headache
Vomiting
Muscle and bone pain
Tiredness
Dry skin
Diarrhea
Nausea
Bleeding
Stomach-area (abdomen) pain
Acne
Talk to your child’s health care provider right away if they experience any side effects. Your child’s health care provider may choose to modify, interrupt, or discontinue treatment.
Here are some helpful tips on how to manage the most commonly seen side effects:
Side Effects | Management Tips |
Fever |
|
Dry skin, rash, or acne | For dry skin, consider:
Contact your child's health care provider if your child gets a skin rash or acne that bothers them or worsens. For mild rash or acne, consider:
|
Headache | Use over-the-counter headache medication to reduce pain |
Upset stomach (including nausea, vomiting, diarrhea, or stomach pain) |
|
Muscle and bone pain |
|
Tiredness |
|
Bleeding | This is a serious problem that requires immediate medical help. Call your child's health care provider right away if you see any signs of bleeding, including:
|
Keep track of your child’s side effects and talk to their health care provider right away about any side effects that are tough to deal with or do not go away. This is important for the health care provider to adjust their treatment as needed. When starting treatment with TAFINLAR + MEKINIST, keep a list of all other medications your child is taking and share it with their health care provider; certain medicines may affect each other, resulting in side effects.
How is TAFINLAR + MEKINIST taken?
TAFINLAR + MEKINIST can be taken in a capsule + tablet form, OR a liquid form. There are specific instructions you will need to follow when giving TAFINLAR + MEKINIST as a liquid—both TAFINLAR and MEKINIST can be given through an oral syringe or feeding tube, and TAFINLAR can also be given in a cup. Our Caregiver's Brochure has additional helpful information, including instructions and tips. Full instructions are available in the Medication Guide for TAFINLAR and Patient Information for MEKINIST.